(C) 2011 Shirley Daniella Martínez-Torres. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
In a Nature Reserve called Reserva Natural Río Nambí (Nariño, Colombia), a platyrhacid millipede population of Psammodesmus bryophorus Hoffman, Martínez & Flórez, 2011 was discovered with 10 epizoic bryophyte species from five families: Fissidentaceae, Lejeuneaceae, Metzgeriaceae, Leucomiaceae and Pilotrichaceae. The inspected sample included 22 Psammodesmus bryophorus individuals of which 15 were carrying mosaics of different bryophyte species on their dorsa, principally Lepidopilum scabrisetum, Lejeunea sp. 1 and Fissidens weirii. This finding constitutes the first record of epizoic plants on Diplopoda.
Diplopoda, Platyrhacidae, Psammodesmus, bryophytes, epizoic, Colombia
Plants that live on living animals are known as epizoic plants. This type of plant has normally been found on tropical vertebrates and is unusual among terrestrial organisms (
Another type of relationship is that of zoochory, only recorded in moss families located in temperate regions: Splachnaceae, whose spores are dispersed by Diptera in Europe (
The present contribution focuses on the first report of epizoic bryophytes on diplopods and the first case of tropical bryophyte entomochory on the backs of diplopods. We show in detail the richness and abundance of epizoic bryophytes on the platyrhacid Psammodesmus bryophorus Hoffman, Martínez and Flórez , 2011 and report three bryophyte families previously unreported as epizoic plants.
MethodsField work was done at the “Reserva Natural Río Nambi” (Fig. 1), located in southwest Colombia near the border with Ecuador, ca 50 km south of Barbacoas and ca 165 km west of Pasto (1°18'N, 78°05'W) at 1100-1900 m (
Geographical location of the Reserva Natural Río Ñambi, borders indicated in red (
Arthropods were sampled by hand, Japanese umbrella device and a Winkler apparatus during two expeditions to the Reserve of 10 days each during October 2009 and May 2010 by biology students under the direction of E. Flórez of the Universidad Nacional de Colombia and the Instituto de Ciencias Naturales. Diplopods were identified to the family level by D. Martínez and E. Flórez following
The two expeditions produced 124 individuals of Diplopoda belonging to six orders and at least 10 families, of which five families were in Polydesmida (Table 1).
Diplopods found in the Reserva Natural Río Ñambí, Colombia.
| Order | Families | Individuals | |
|---|---|---|---|
| Polydesmida | Platyrhacidae | 56 | |
| Chelodesmidae | 6 | ||
| Aphelidesmidae | 4 | ||
| Cryptodesmidae | 1 | ||
| Cyrtodesmidae | 1 | ||
| Spirobolida | Rhinocricidae | 17 | |
| Not determined | 1 | ||
| Spirostreptida | Spirostreptidae | 1 | |
| Stemmiulida | Stemmiulidae | 15 | |
| Glomeridesmida | Glomeridesmidae | 19 | |
| Siphonophorida | Siphonophoridae | 3 | |
| Total | 6 | 10 + | 124 |
During the process of sorting and identifying the diplopods, it was found that several specimens of Psammodesmus bryophorus (Polydesmida, Platyrhacidae), recently described by
Bryophyte-diplopod associations.
| Psammodesmus bryophorus | With bryophytes | Without bryophytes | Total |
|---|---|---|---|
| Males | 14 | 6 | 20 |
| Females | 1 | 1 | 2 |
Bryophytes found on 15 specimens of Psammodesmus bryophorus
| Family | Species | Habit | Individual plants | |
|---|---|---|---|---|
| Pilotrichaceae | Lepidopilum scabrisetum (Schwägr.) Steere | Epiphyllous | 247 | |
| Lejeuneaceae | Lejeunea sp. 1 | Epiphyllous | 75 | |
| Fissidentaceae | Fissidens weirii Mitt. | Edaphic | 34 | |
| Lejeuneaceae | Drepanolejeunea sp. 1 | Epiphyllous | 16 | |
| Fissidentaceae | Fissidens steerei Grout | Edaphic | 14 | |
| Lejeuneaceae | Cyclolejeunea | Epiphyllous | 5 | |
| Metzgeriaceae | Metzgeria sp. | Epiphyllous | 3 | |
| Lejeuneaceae | Lejeunea sp. 2 | Epiphyllous | 3 | |
| Lejeuneaceae | Drepanolejeunea sp. 2 | Epiphyllous | 2 | |
| Leucomiaceae | Leucomium strumosum (Hornsch.) Mitt. | Epiphyllous | 2 | |
| Total | 5 | 10 | 2 | 401 |
The most abundant bryophyte families are shown in Fig. 2.
Examples of bryophytes on Psammodesmus bryophorus. From right to left: Lejeuneaceae, Fissidentaceae, Pilotrichaceae.
Most of the individuals of Psammodesmus bryophorus were found on tree trunks and living leaves, about 1 m above the ground, while some were found between litter and the soil surface.
Bryophytes were discovered on most parts of the body (Fig. 3) except the head. Ninety percent were found on the back, with the majority located on the keels, although some were found on the middle of the metatergite. Only six percent were found on the ventral side, towards the base of the legs.
Bryophytes on a Psammodesmus bryophorus male.
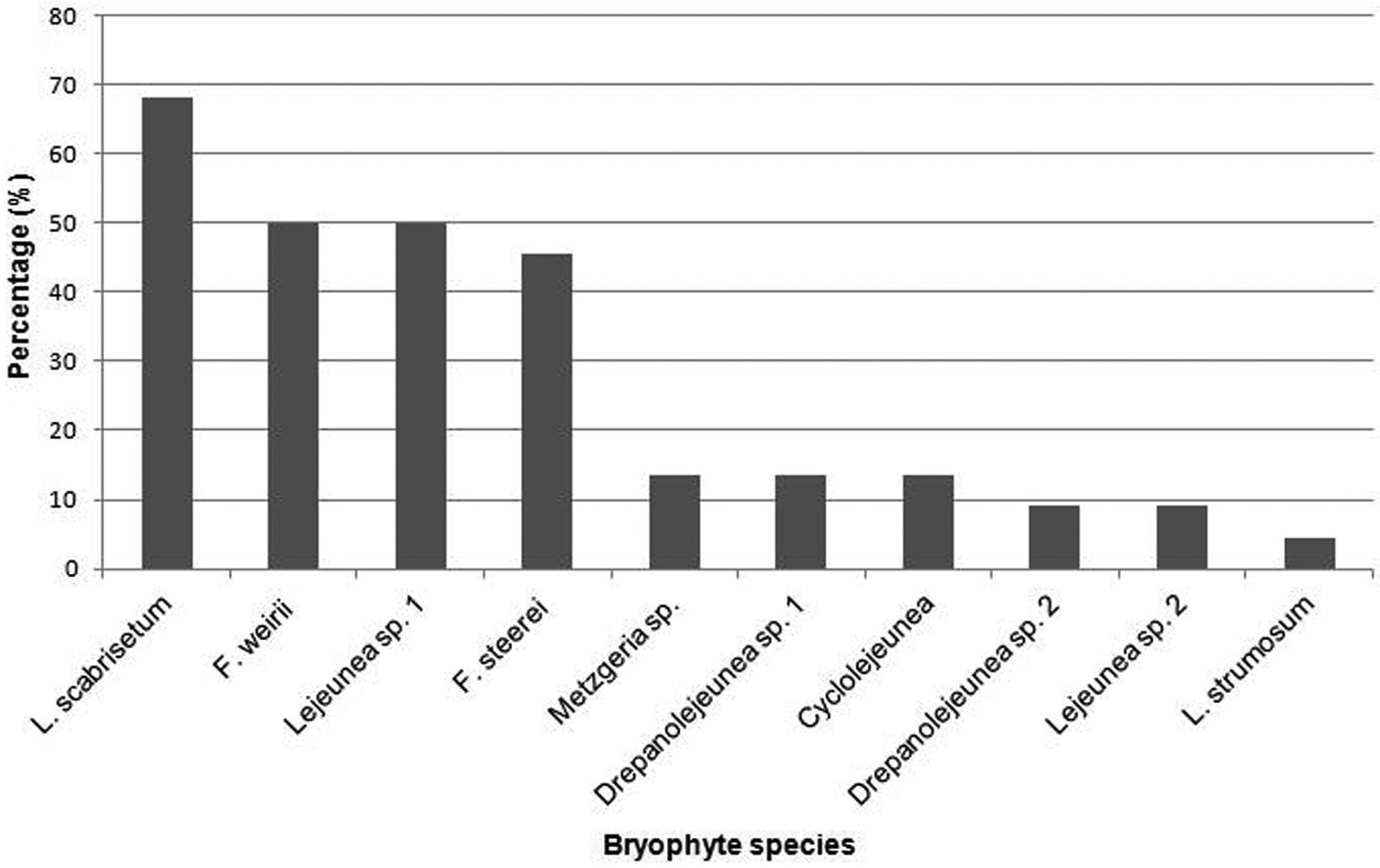
Lepidopilum scabrisetum was not only the most abundant bryophyte species on Psammodesmus bryophorus, but also the species found in the majority of millipede individuals, being present in 68%, followed by Fissidens weirii and Leujenea sp. 1, which are present in 50% of individuals (Fig. 4). Leucomium strumosum was only found on one individual.
Presence percentage of bryophyte species on Psammodesmus bryophorus.
A male had the highest abundance of bryophytes (55 individual plants) as well as the greatest diversity (seven species) followed by three males with six bryophyte species and a mean abundance of 40 individuals.
DiscussionBryophytes do not establish easily on unstable substrates with high decomposition rates, such as litter and soil surfaces in the Reserva forest. It may be that Psammodesmus bryophorus cuticle serves as a more stable substrate and thus allows the bryophytes to persist and disperse in an otherwise difficult environment. They may also provide a benefit to Psammodesmus bryophorus, which is darkly colored dorsally with two longitudinal light stripes; the epizoic bryophytes may camouflage the diplopod and help protect it from predation as has been reported for other arthropods (
The Leucomiaceae and Metzgeriaceae families found in this work have already been reported as epizoic bryophytes (
We have reported here the first known associations of bryophytes with Diplopoda, and one of the few with Arthropoda. It is also, so far as we know, the first reported case of tropical bryophyte entomochory, in which spores and propagules that fell on the backs of diplopods germinated and produced small plants. Most of the epizoic bryophytes we identified are normally epiphytic, but two are normally ground-based near watercourses. Finally, none of the previous studies reported multiple species of bryophytes on the same arthropod species, much less on the same individual as found in this study. This may be related to the size of Psammodesmus bryophorus, which provides a considerable area of bryophyte grown substrate on its body surface.
We thank Dr R. Hoffman, Curator of Invertebrates at the Virginia Museum of Natural History, Martinsville, Virginia, USA, for his assistance with Psammodesmus bryophorus, Mauricio and Cristian Florez of FELCA for their help with the expeditions and all those who directly or indirectly contributed to this study.